Are All Molecular Compounds Soluble In Water
Some types of ionic compounds however cannot be written with entirely whole-number. 1Most PEGs are commonly available commercially as mixtures of different oligomer sizes in broadly- or narrowly-defined molecular weight MW ranges.

Dissolving Process Chemistry For Non Majors
For example the empirical formula of ethanol may be written C 2 H 6 O because the molecules of ethanol all contain two carbon atoms six hydrogen atoms and one oxygen atom.
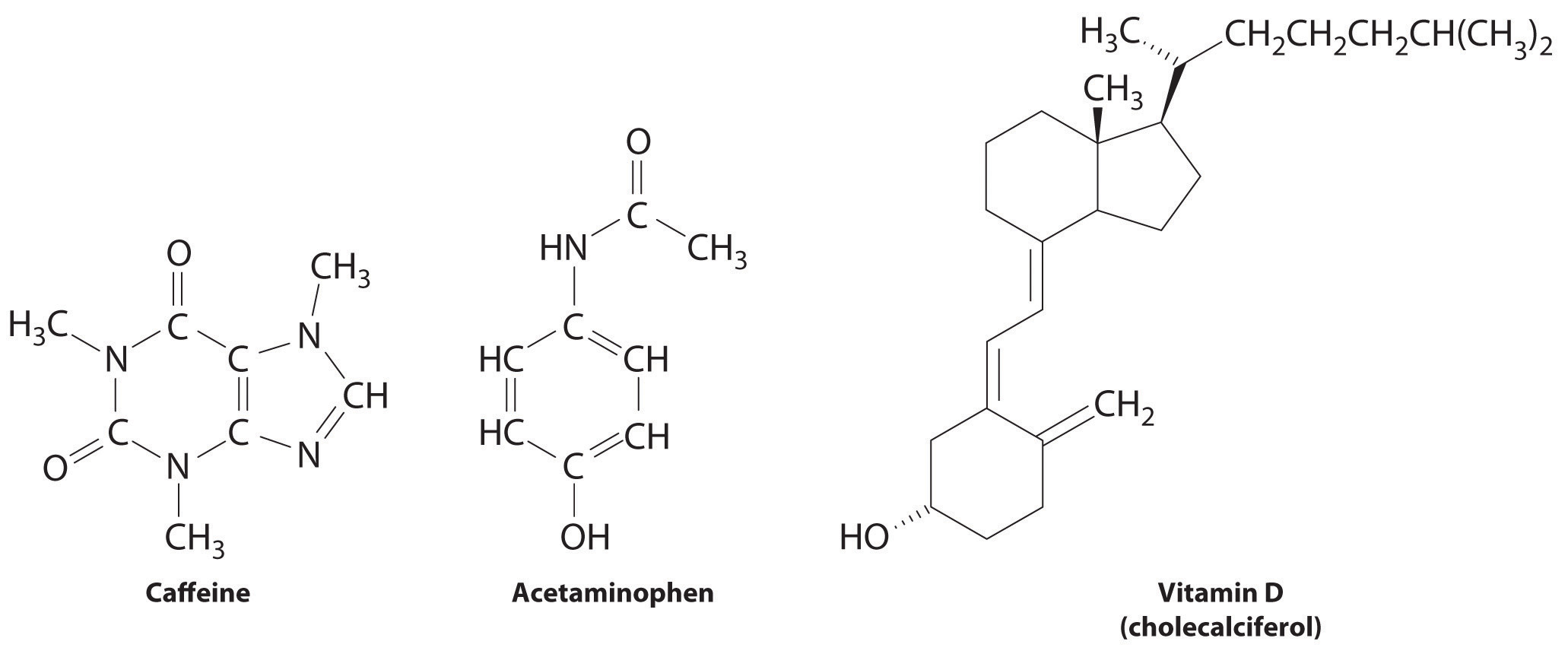
Are all molecular compounds soluble in water. But benzoic acid is not soluble in water. Polyethylene glycols PEGs are composed of polyether compounds repeating ethylene glycol units according to the constituent monomer or parent molecule as ethylene glycol ethylene oxide or oxyethylene Fig. Fatty acids made up of ten or more carbon atoms are nearly insoluble in water and because.
Carboxylic acids and salts having alkyl chains longer than eight carbons exhibit unusual behavior in water due to the presence of both hydrophilic CO 2 and hydrophobic alkyl regions in the same moleculeSuch molecules are termed amphiphilic Gk. Sodium acetate is also used in heating pads hand warmers and hot iceSodium acetate trihydrate crystals melt at 1364 F58 C to 13712 F584 C dissolving in their water of crystallizationWhen they are heated past the melting point and subsequently allowed to cool the aqueous solution becomes supersaturatedThis solution is capable of cooling to room temperature without forming. Both of compounds are carboxylic acids.
Atoms are a lot like us - we call their relationships bonds and there are many different types. Each kind of atomic relationship requires a different type. Alcohols carboxylic acids carboxylic acid chlorides amines esters are usually soluble in water.
All CrVI-containing compounds were once thought to be man-made with only CrIII naturally ubiquitous in air water soil and biological materials. Amphi both or amphipathic. As examples ethanoic acid is soluble in water.
But when those compounds molecular mass increases solubility in water is decreased. Recently however naturally occurring CrVI has been found in ground and surface waters at values exceeding the World Health Organization limit for drinking water of 50 µg of CrVI per liter. For molecular compounds these ratio numbers can all be expressed as whole numbers.

Properties Of Solutions Ppt Video Online Download
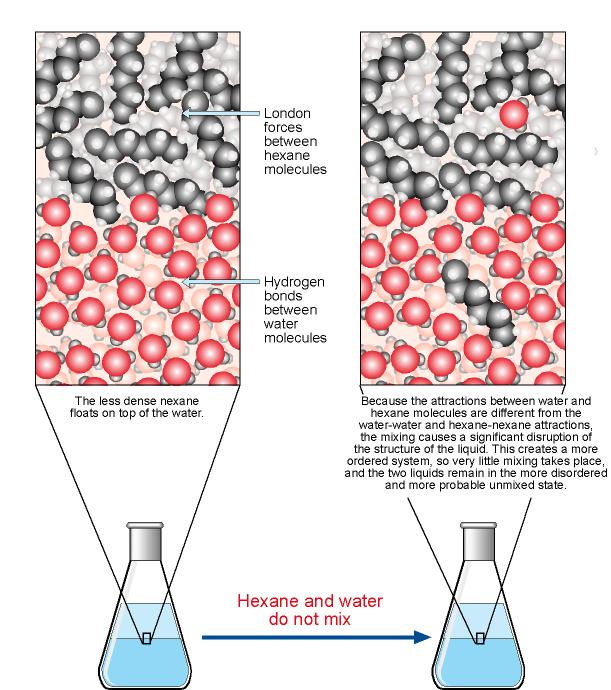
Solubility And Molecular Structure

Chemistry Ii Water And Organic Molecules

Chemistry Ii Water And Organic Molecules

Chemistry Ii Water And Organic Molecules

Chemistry Ii Water And Organic Molecules
What Determines Whether A Solid Is Soluble In Water Socratic
What Kind Of Compound Would Be Least Likely To Dissolve In Water Socratic
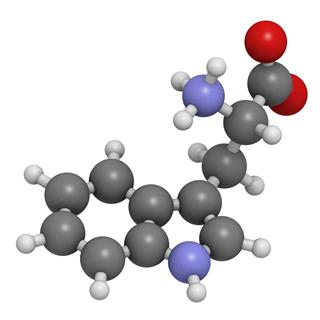
Ionic Compounds Vs Molecular Compounds What You Need To Know Science Struck

Chemistry Ii Water And Organic Molecules

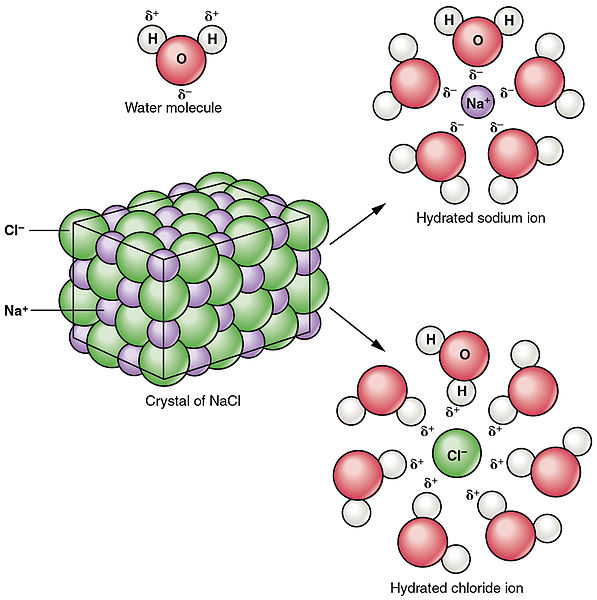
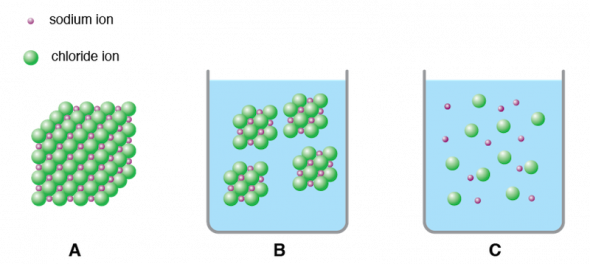
Water Molecules And Their Interaction With Salt
6 2 Comparing Ionic And Molecular Substances Chemistry Libretexts

Sort The Following Compounds Based On Whet Clutch Prep
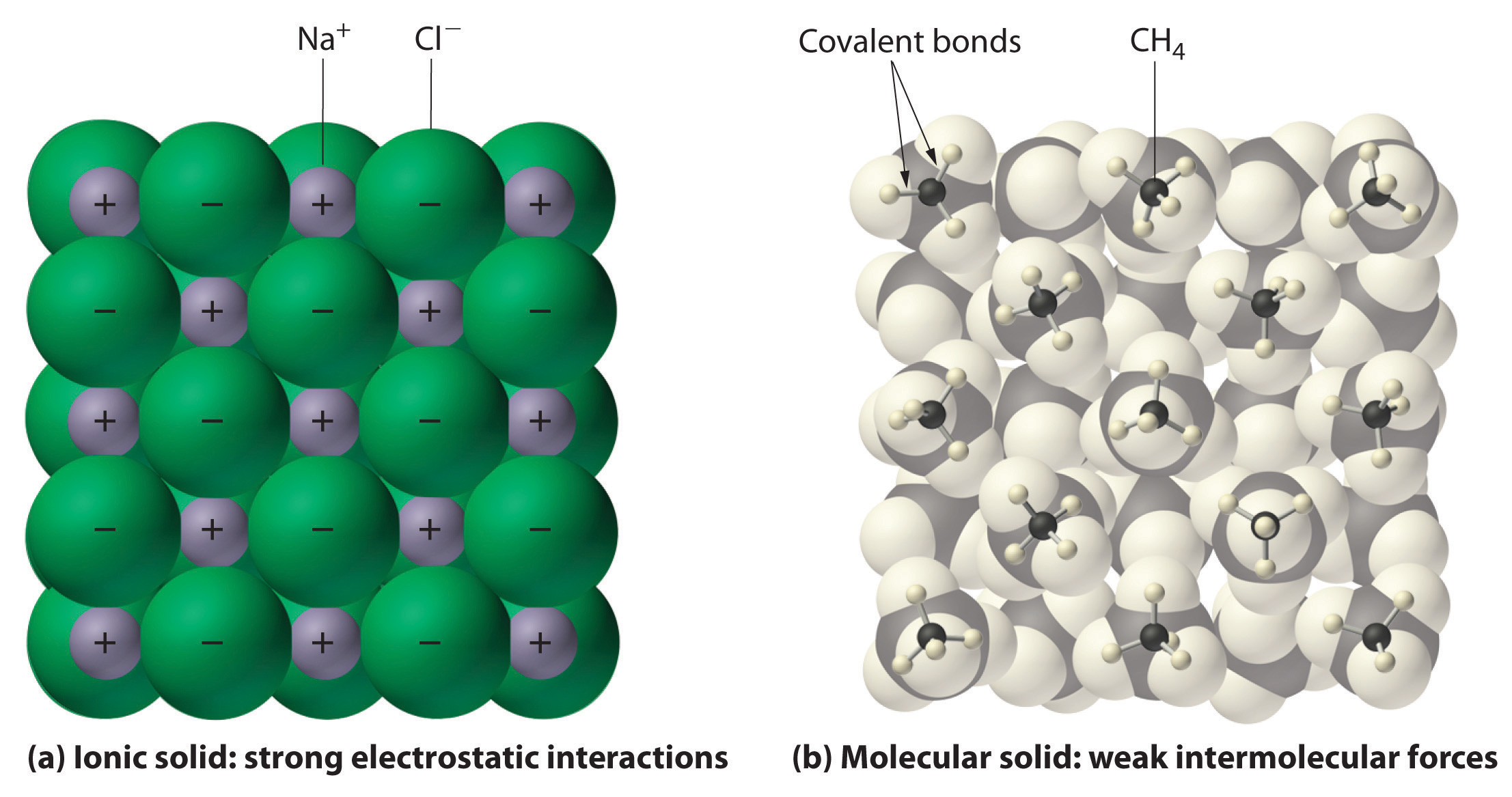
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Solubility And Dissolving Vce Chemistry
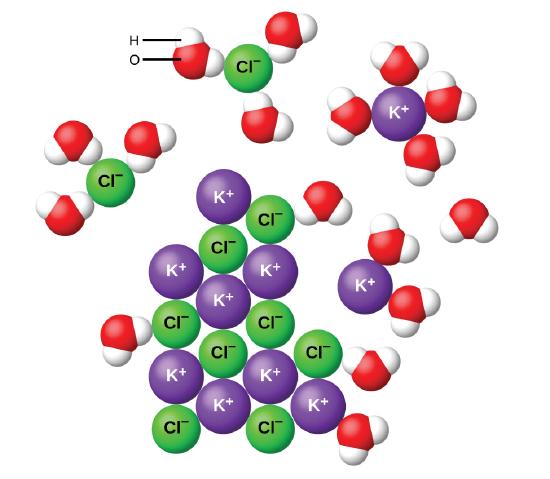
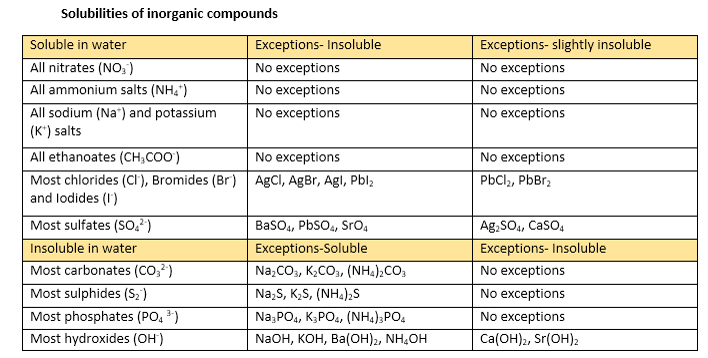
7 5 Aqueous Solutions And Solubility Compounds Dissolved In Water Chemistry Libretexts
Covalent Compounds Manoa Hawaii Edu Exploringourfluidearth