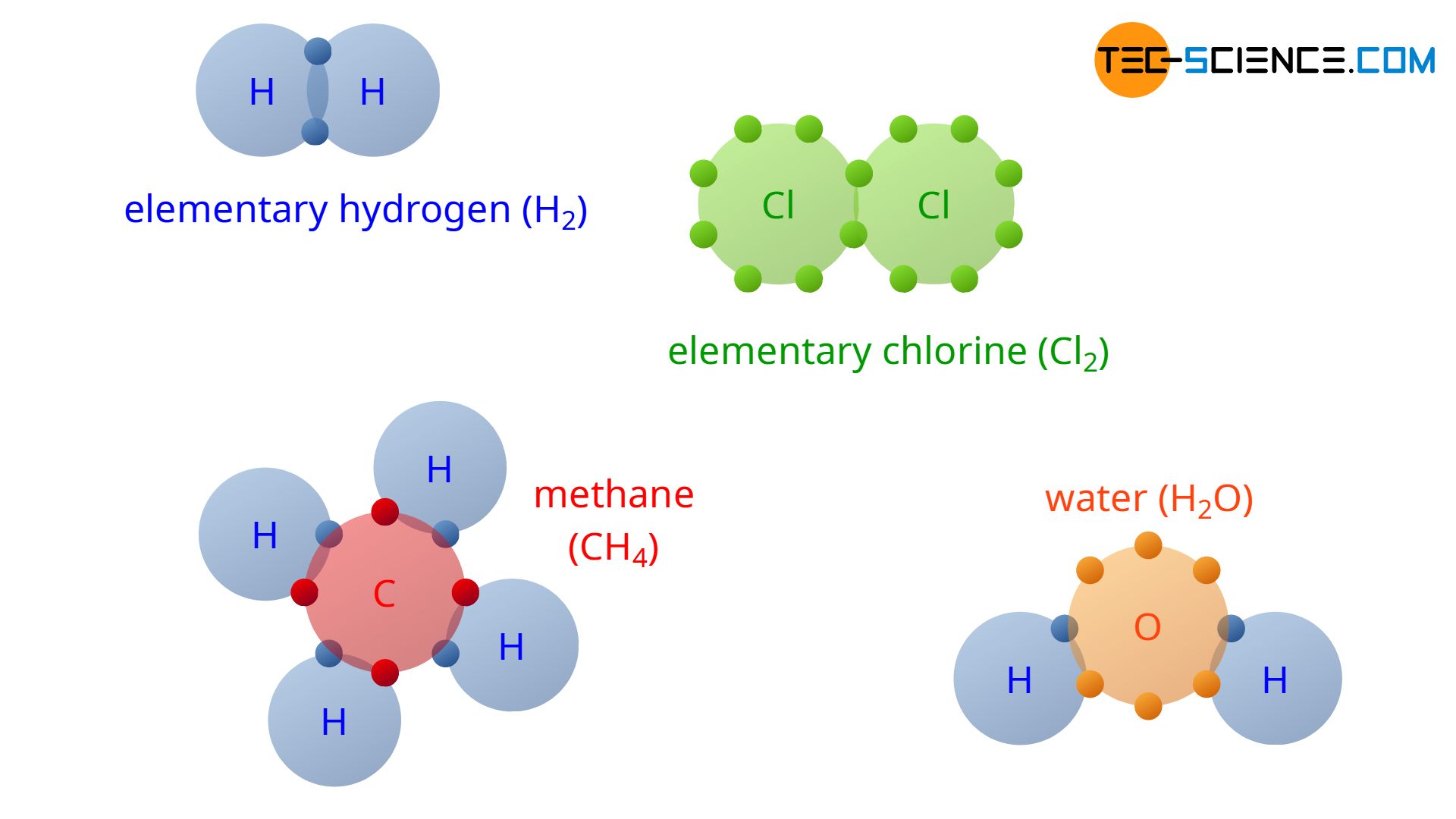
What Are Covalently Bonded Compounds Called
The C 6 F 5XeF 2 cation has been prepared as the BF 4 salt. Each mercury atom within the bonded pair has a charge state of 1.
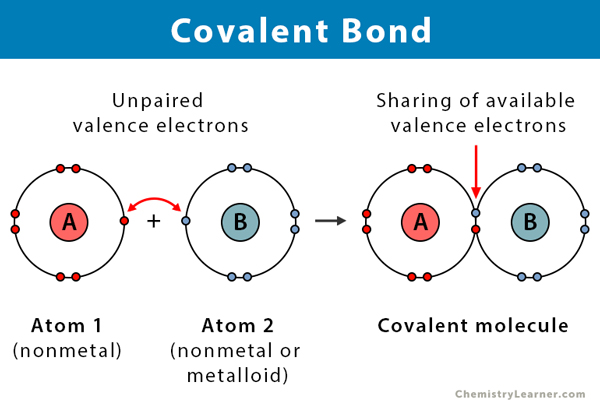
Covalent Bond Definition Types And Examples
Some covalently bounded compounds have a small difference in charge along one direction of the molecule.
What are covalently bonded compounds called. An example of xenon4 bonded to carbon is also known. Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons in the form of an electron cloud of delocalized electrons and positively charged metal ionsIt may be described as the sharing of free electrons among a structure of positively charged ions Metallic bonding accounts for many physical properties of metals such as. Covalent bonds are formed as a result of the sharing of one or more pairs of bonding electrons.
These kinds of bonds occur when the shared electrons are not shared equally between atoms. Ketone any of a class of organic compounds characterized by the presence of a carbonyl group in which the carbon atom is covalently bonded to an oxygen atom. Carbon dioxide or CO 2 is a naturally occurring linear compound composed of two oxygen atoms covalently bonded to a carbon atom.
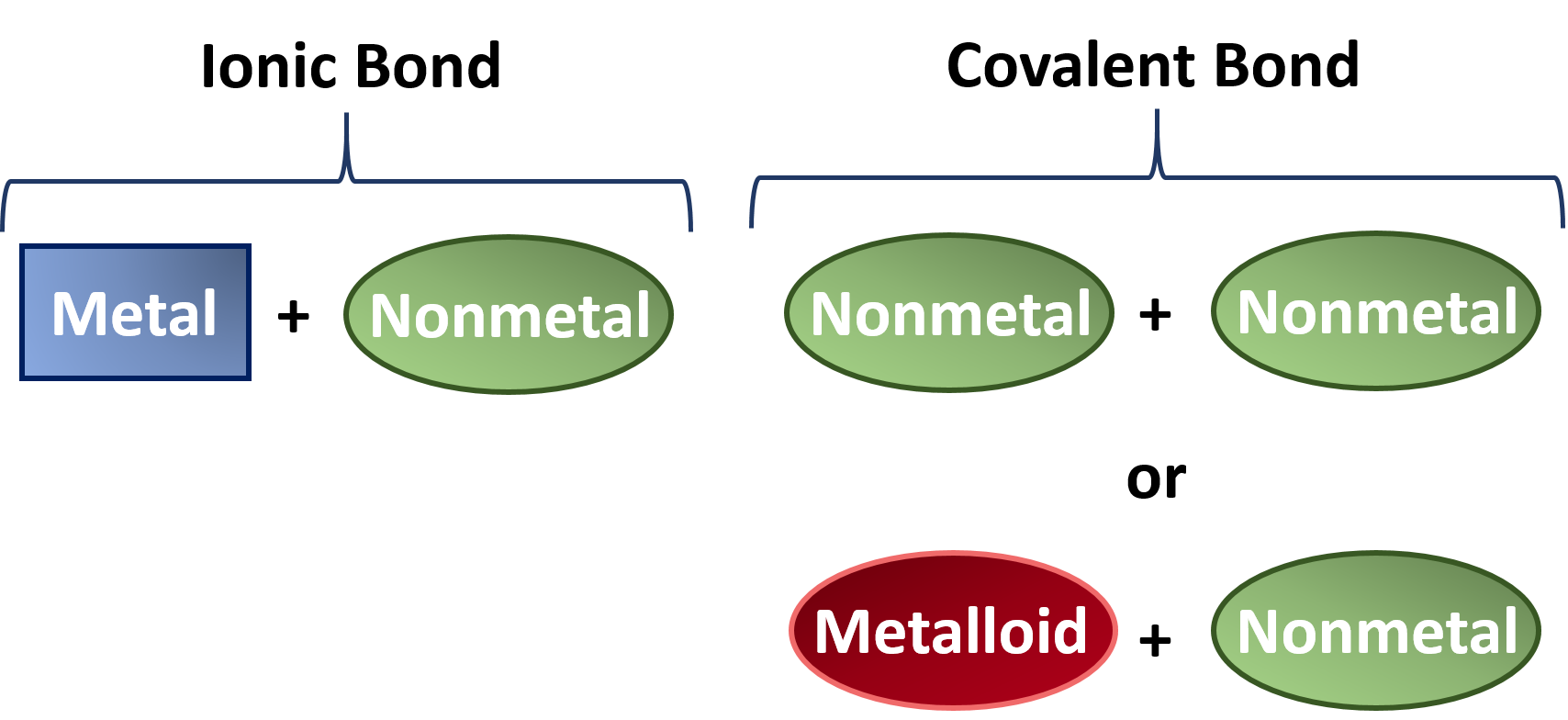
The remaining two bonds are to other carbon atoms or hydrocarbon radicals. The resulting compounds are called ionic compounds. This difference in charge is called a dipole and when the covalent bond results in this difference in charge the bond is called a polar covalent bond.
The most important ketone is acetone a liquid with a sweetish odor. The two CO bonds are equivalent and short 1163 pm consistent with double bonding. As long as the electro-negativity difference is no greater than 17 the atoms can only share the bonding electrons.
This give the overall ion a 2 state as shown below. A number of compounds containing Xe-C bonds are known. These compounds are salts of cations containing xenon2 coordinated to carbon and include cations such as C 6 F 5Xe and m-CF 3 C 6 H 4Xe.
The electro negativities electron attracting ability of the two bonded atoms are either equal or the difference is no greater than 17. Hg 2 2 where two atoms of mercury are actually covalently bonded to one another as a polyatomic ion.
What Is A Covalent Bond How Many Types Of Covalent Bond Are There Quora
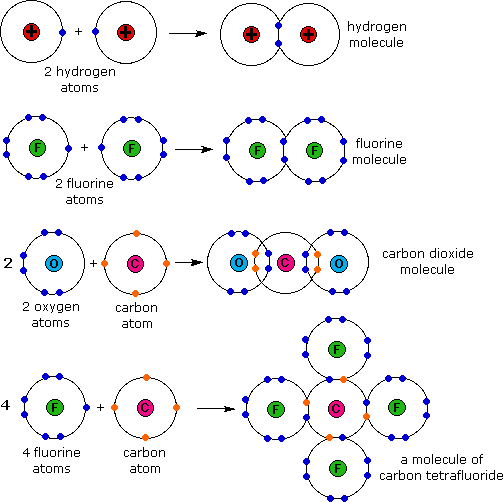
As Studypeach Covalent Bonds Sharing Is Caring

Covalent Bond Examples Formation Properties What Is A Covalent Bond Video Lesson Transcript Study Com

Covalent Bonding Biology Definition Role Expii
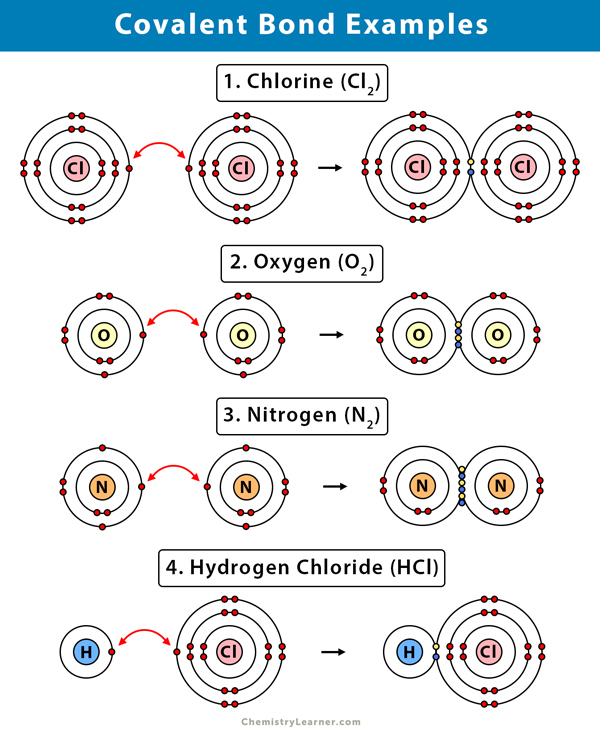
Covalent Bond Definition Types And Examples
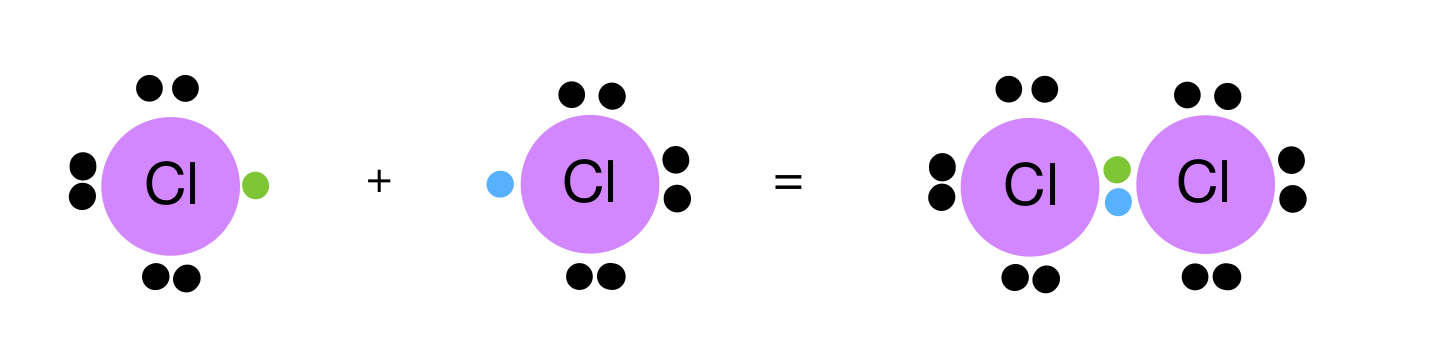
Covalent Bond Formation Compounds Expii

Covalent Bond An Overview Sciencedirect Topics
What Does A Covalent Bond Mean Quora
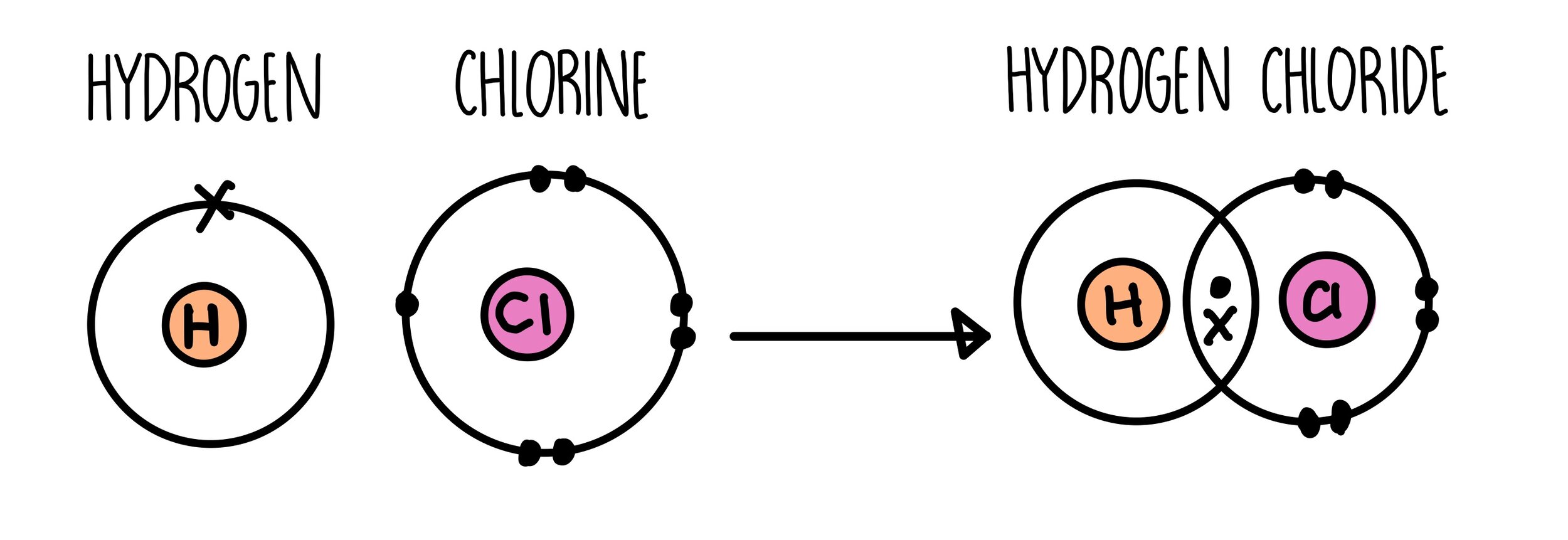
Covalent Bonding Gcse The Science Hive

Covalent Bond Examples Science Struck

Covalent Bond Definition Properties Examples Facts Britannica

Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
Covalent Bond Definition Types Polar And Non Polar Covalent Bond Explained

Naming Covalent Compounds Nomenclature Rules
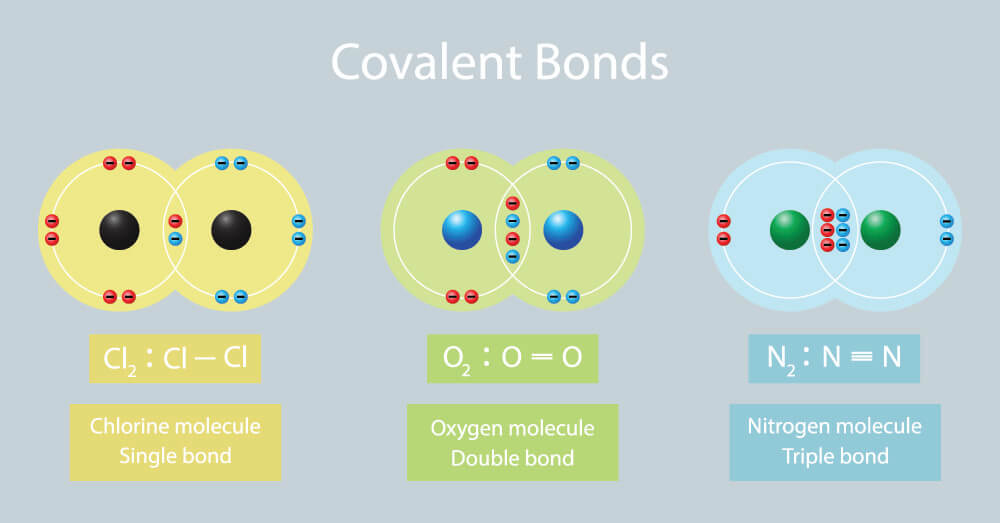
Covalent Bond Biology Dictionary

Covalent Bonds Biology For Majors I

Covalent Bond An Overview Sciencedirect Topics

Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry